WELCOME TO HEAD & NECK ROBOTIC SURGERY IN THE H&N
The OTTAVA™ Robotic Surgical System is Johnson & Johnson (J&J) MedTech’s next‑generation platform for soft‑tissue surgery. Designed primarily for complex general surgery procedures, OTTAVA represents the culmination of more than a decade of development in surgical robotics, digital surgery, and minimally invasive instrumentation within the J&J ecosystem.
As of Jan 2026, OTTAVA has been submitted to the U.S. Food and Drug Administration (FDA) for de novo classification and is not yet commercially available.
Chronological History
J&J’s modern robotics strategy began in 2015 with the creation of Verb Surgical, a joint venture between Ethicon (J&J) and Verily Life Sciences (Alphabet/Google). The ambition was bold: to create a digitally enabled surgical platform combining robotics, advanced instrumentation, data analytics, connectivity, and machine learning.
Verb Surgical operated largely in secrecy, but public statements emphasized a future of data‑driven, connected surgery, rather than robotics as a stand-alone tool.
In 2019, Johnson & Johnson acquired Auris Health, founded by Dr. Frederic Moll, co‑founder of Intuitive Surgical. Auris brought with it the Monarch™ robotic platform, primarily focused on endoluminal procedures (neumonology and urology).
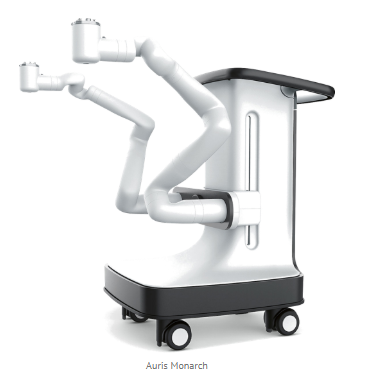
Later that year, J&J bought out Verily’s stake in Verb Surgical, consolidating full ownership of its robotics programs. Technologies, teams, and concepts from Verb Surgical and Auris Health were progressively integrated, laying the groundwork for a unified soft‑tissue robotic platform.
In 2020, J&J publicly introduced the name OTTAVA™, derived from the Italian word for “octave,” reflecting harmony, coordination, and precision. Early disclosures highlighted a novel concept: a robotic system integrated directly into the operating table, rather than mounted on external carts.
Ottava : Is this how the arms and workflow work? Steve Bell Surgical Robotics.
This architectural decision signaled a clear departure from existing systems, with a focus on:
Between 2021 and 2022, J&J acknowledged development delays, extending the anticipated clinical timeline by approximately two years. These delays reflected the technical complexity of integrating robotics, table motion, instrumentation, and digital systems into a single platform, as well as broader industry disruptions during the COVID‑19 period.
Despite these challenges, the company reaffirmed its long‑term commitment to OTTAVA as a cornerstone of its surgical future.
In late 2024, OTTAVA received Investigational Device Exemption (IDE) approval from the FDA, enabling the start of U.S. clinical trials. This marked the transition from preclinical development to human use in controlled clinical settings. The IDE allowed evaluation of OTTAVA in complex general surgery procedures, particularly in the upper abdomen.
In 2025, the first clinical cases using OTTAVA were performed at U.S. centers, including Y-Roux gastric bypass procedures. These cases formed part of the structured IDE clinical study and provided the initial human safety and performance data required for regulatory submission.
In January 2026, Johnson & Johnson announced the submission of the OTTAVA Robotic Surgical System to the FDA for De Novo classification. The application is based on data from the IDE clinical study and targets indications in soft‑tissue general surgery, particularly upper‑abdominal procedures.
The OTTAVA System: Technical Overview
OTTAVA’s defining feature is its full integration into the operating table:
The design minimizes clutter and maximizes access for anesthesia, assistants, and additional equipment
The system allows synchronized movement of the patient table and robotic arms, enabling:
OTTAVA is designed to support:
This reflects J&J’s philosophy of supporting the surgeon’s workflow, rather than forcing procedural rigidity.
The system leverages Ethicon surgical instrumentation, drawing on decades of experience in laparoscopic and open surgery. The goal is to provide familiar performance characteristics while extending precision and control through robotic assistance.

Digital Ecosystem: Polyphonic™
OTTAVA is intended to operate within J&J’s broader Polyphonic™ Digital Ecosystem, a developing platform designed to connect:
This ecosystem approach aligns with an industry‑wide trend toward integrated, data‑centric surgical platforms.
The current regulatory submission focuses on soft‑tissue general surgery, including complex upper‑abdominal procedures such as gastric bypass and bariatric surgery, small bowel and gastric procedures, and hiatal and ventral hernia repair. Future expansions to other specialties will depend on regulatory outcomes and clinical strategy.
OTTAVA represents J&J’s most comprehensive attempt to enter the soft‑tissue surgical robotics market with a fully integrated, ecosystem‑based platform. Its table‑mounted design, flexible workflow philosophy, and deep integration with Ethicon instrumentation distinguish it from earlier generations of surgical robots.
This page will be updated as new regulatory, clinical, and technical information becomes available.
– Johnson&Johnson Surgical Robotics
OTTAVA (J&J´s Robotic Surgical System) Moves Closer to Market Jan 8, 2025